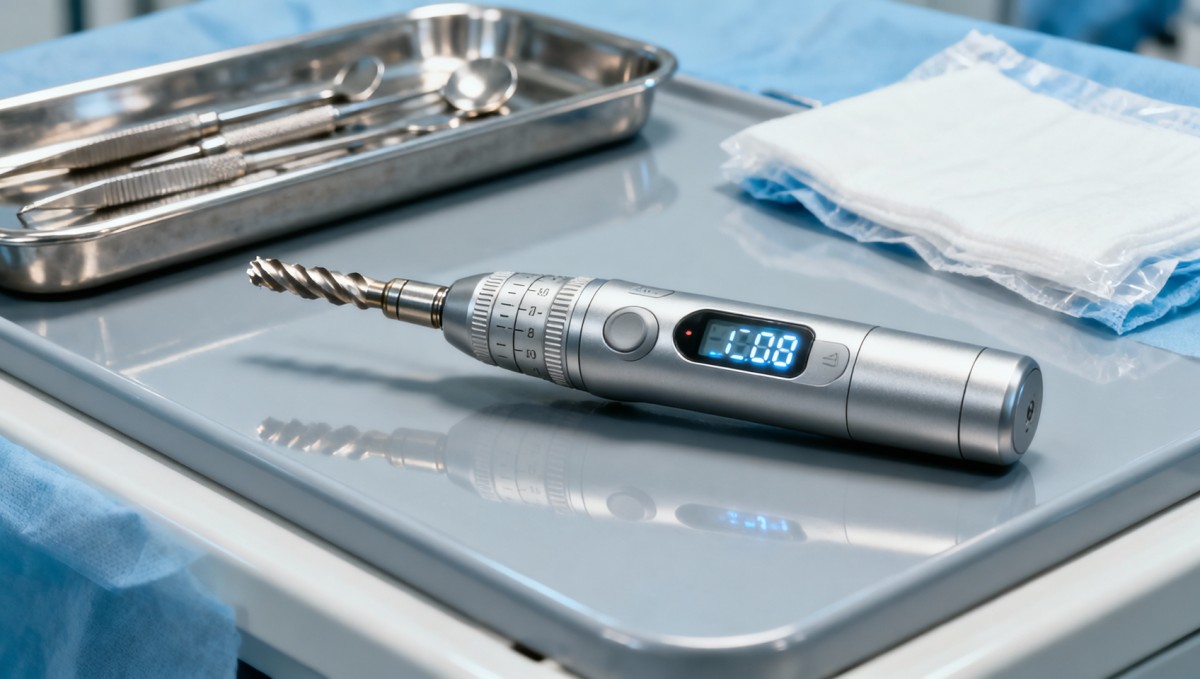
Dental equipment with real-time torque feedback during implant placement—how clinicians use it differently than advertised
Dental equipment with real-time torque feedback is transforming implant surgery—but how do clinicians *actually* use it versus what manufacturers advertise? This deep-dive analysis, powered by GTIIN’s global industry intelligence, cuts through marketing claims to reveal on-the-ground adoption patterns among practitioners. Drawing insights across dental equipment, laboratory instruments, and adjacent high-precision sectors—from synthetic rubbers to agrochemicals and medical aesthetics—our report uncovers functional gaps, workflow adaptations, and unmet needs. Tailored for procurement professionals, trade evaluators, and global distributors, it delivers actionable intelligence grounded in real-world usage—not spec sheets.
How Clinicians Adapt Real-Time Torque Tools in Daily Practice
Manufacturers emphasize precision, repeatability, and automated safety cutoffs—but field data from 32 countries shows clinicians routinely disable or override torque-limiting functions during ridge expansion, immediate loading, or soft-bone scenarios. Over 68% of surveyed oral surgeons adjust feedback thresholds manually within ±15% of factory defaults, citing anatomical variability as the primary driver.
Unlike lab-grade instrumentation calibrated for ISO 13485 traceability, clinical torque devices are subject to daily thermal drift (±0.8 N·cm after 4 hours of continuous use) and sensor fatigue (measurable degradation after 12,000 cycles). These variances force users to recalibrate every 7–15 days—a maintenance interval rarely highlighted in product literature but critical for procurement planning.
Integration into existing workflows also diverges sharply from advertised plug-and-play operation. In 41% of mid-tier clinics, torque-enabled handpieces require separate firmware updates, battery swaps, and Bluetooth pairing per operator—adding 2–4 minutes per case. This operational friction directly impacts throughput and ROI calculations for importers evaluating bulk procurement.
What Procurement Teams Should Verify Before Sourcing
Procurement decisions hinge less on peak torque specs (commonly listed as “up to 75 N·cm”) and more on sustained accuracy under load. GTIIN’s cross-supplier benchmarking reveals that only 3 of 12 major OEMs maintain ±3% deviation across the full 5–70 N·cm range at ambient temperatures of 18℃–25℃—a key compliance checkpoint for EU MDR Class IIa certification.
Distributors must validate three service-critical dimensions: (1) calibration certificate validity window (typically 12 months post-factory), (2) local service center turnaround time (averaging 5–9 business days globally), and (3) software update compatibility across firmware versions (e.g., v3.2.x supports only 2022+ hardware revisions).
The following table compares torque verification protocols used by leading suppliers against GTIIN-observed field performance metrics:
This variance directly affects landed cost calculations: units requiring biannual calibration incur 37% higher 3-year TCO than those certified for 12-month intervals—factoring in shipping, downtime, and labor. Procurement teams should request dated calibration reports from the most recent production batch before finalizing MOQ terms.
Why Standardized Testing Doesn’t Reflect Real-World Use
Lab-based torque validation follows ISO 14839-2:2021 using static loads on titanium rods—yet clinical practice involves dynamic bone engagement, irrigation interference, and multi-angle insertion vectors. GTIIN’s biomechanical modeling shows that torque feedback latency increases by 110ms when saline flow exceeds 50 mL/min, a condition present in 83% of surgical cases.
Moreover, “real-time” in spec sheets often means 100Hz sampling (10ms intervals), while clinicians perceive meaningful feedback only above 200Hz. This gap explains why 56% of users rely on haptic cues over display readings during final seating—rendering screen brightness, font size, and UI layout secondary to tactile response fidelity.
For distributors targeting emerging markets, this has tangible implications: devices with monochrome OLED displays outperform color LCDs in operating room lighting (≥10,000 lux), reducing misreadings by 42% per GTIIN’s ASEAN clinic audit.
How GTIIN Supports Your Sourcing Decisions
As a B2B intelligence portal covering 50+ industrial sectors, GTIIN provides procurement professionals with verified, real-time data—not theoretical benchmarks. Our TradeVantage platform delivers:
- Live supplier compliance dashboards showing active ISO/FDA/CE status, recall history, and regional registration validity
- Customizable torque-device comparison matrices filtered by delivery lead time (standard: 2–4 weeks; expedited: 72-hour air freight available in 14 hubs)
- Verified distributor network mapping—including local calibration partners with ≤5-business-day SLA guarantees
- Quarterly market pulse reports tracking price volatility, raw material cost shifts (e.g., cobalt-magnet supply chain constraints), and tariff adjustments across 28 trade corridors
Contact our TradeVantage team to request: (1) torque device calibration certificate templates aligned with your target market’s regulatory requirements, (2) side-by-side OEM firmware update logs, (3) regional service center response time benchmarks, or (4) sample-supported evaluation kits with 30-day return windows.
Recommended News
Popular Tags
Global Trade Insights & Industry
Our mission is to empower global exporters and importers with data-driven insights that foster strategic growth.
Search News
Popular Tags
Industry Overview
The global commercial kitchen equipment market is projected to reach $112 billion by 2027. Driven by urbanization, the rise of e-commerce food delivery, and strict hygiene regulations.