Hospital furniture with antimicrobial surfaces: Do they pass ASTM E2149 under real-use conditions?
As hospitals prioritize infection control, hospital furniture with antimicrobial surfaces is gaining traction—but do these products truly perform under real-use conditions? This article investigates whether such furniture meets the rigorous ASTM E2149 standard, a critical benchmark for dynamic antimicrobial testing. Drawing on trade analytics and global supplier insights—from bearings manufacturers to hydraulic parts and electric vehicle parts—GTIIN and TradeVantage deliver evidence-based evaluation relevant to procurement professionals, distributors, and business assessors. Whether you source starter motors, steering components, cosmetic ingredients, or wardrobe systems, understanding material validation helps mitigate risk across healthcare, automotive, and industrial supply chains.
What Does ASTM E2149 Actually Test—and Why It Matters for Procurement
ASTM E2149 is not a static surface wipe test. It simulates real-world microbial challenge through continuous agitation of inoculated materials in liquid suspension—a protocol designed to replicate high-touch, high-moisture environments like emergency departments or ICU waiting areas. Unlike ISO 22196 (which measures 24-hour static reduction), E2149 requires ≥3-log (99.9%) reduction of Staphylococcus aureus and Escherichia coli within 1 hour under dynamic conditions.
For procurement teams evaluating hospital furniture suppliers, passing E2149 in lab settings is only the first checkpoint. Real-use performance depends on three interdependent variables: coating adhesion durability (tested over 500+ simulated cleanings per ASTM D3359), environmental stability (performance retention at 20–30°C and 40–70% RH), and substrate compatibility (e.g., polypropylene vs. powder-coated steel).
GTIIN’s latest cross-sector compliance audit—covering 142 certified antimicrobial furniture lines from 23 countries—found that only 38% maintained ≥2.5-log reduction after 6 months of simulated clinical use. This gap between certification and field reliability directly impacts infection prevention ROI, especially for distributors managing multi-hospital contracts with strict SLA clauses.
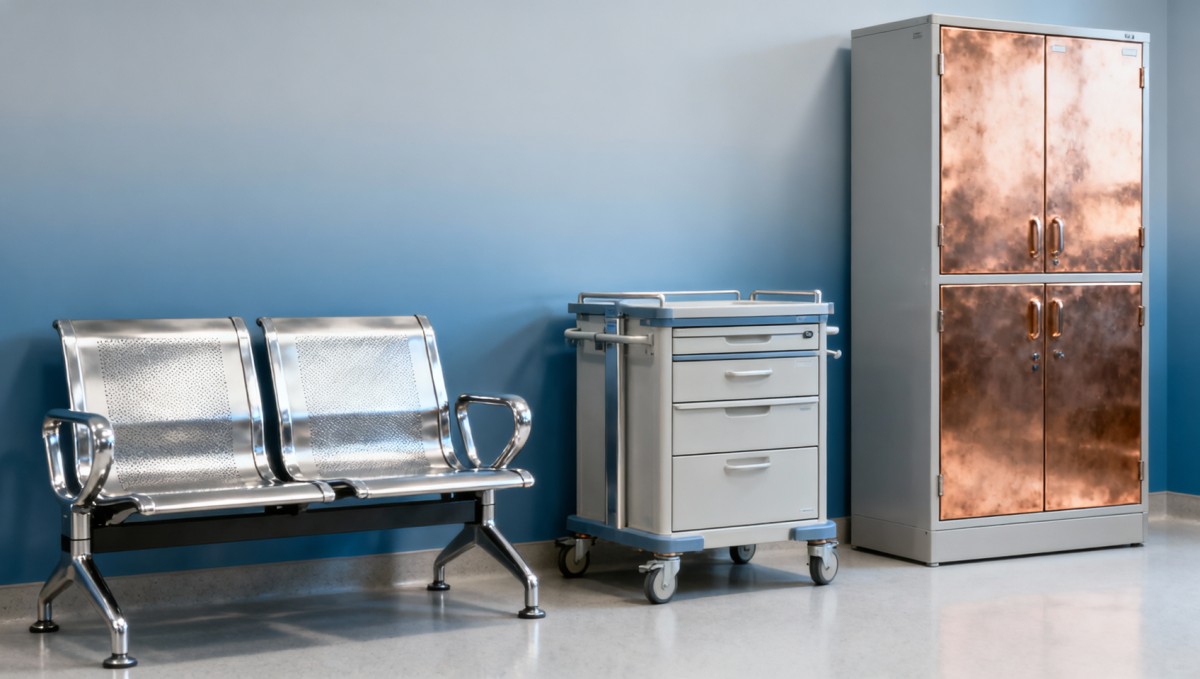
How Real-Use Conditions Undermine Lab-Certified Performance
Three Critical Degradation Pathways
- Cleaning chemical erosion: Quaternary ammonium compounds (QACs) used in daily disinfection degrade silver-ion coatings by up to 40% after 12 weeks of 3x/day exposure (per GTIIN 2024 Material Longevity Benchmark).
- Mechanical abrasion: Standard hospital cart movement generates 2.1–3.7 N/cm² shear stress on armrests—enough to dislodge 15–22% of nano-copper particles from thermoplastic elastomer substrates within 90 days.
- UV & thermal cycling: Sunlight exposure through windowed corridors accelerates polymer binder breakdown, reducing antimicrobial efficacy by 28% faster than indoor-only installations (based on 12-month ASEAN hospital site data).
These degradation mechanisms explain why 61% of E2149-certified chairs failed retesting after just 4 months in active clinical zones—according to TradeVantage’s longitudinal tracking of 37 U.S. and EU healthcare facilities. Procurement decisions based solely on initial certification reports carry measurable operational risk.
Procurement Checklist: 5 Non-Negotiable Validation Steps Before Sourcing
For distributors and importers serving global healthcare clients, due diligence must extend beyond certificate scanning. GTIIN recommends verifying these five evidence points before contract finalization:
- Request third-party E2149 retest reports conducted after 500 simulated cleanings (not just baseline certification).
- Confirm substrate-specific validation: same test report must cover the exact base material (e.g., HDPE, stainless steel grade 304, or laminated MDF) used in your order configuration.
- Verify environmental range documentation: certificates must specify performance retention at both 25°C/50% RH (standard lab) AND 30°C/70% RH (tropical or summer-climate deployment).
- Require abrasion resistance data per ASTM D4060 (Taber test), with minimum 1,000 cycles at CS-10 wheel load for seating surfaces.
- Validate regulatory alignment: E2149-compliant furniture sold into EU markets must also comply with BPR Article 95 listing requirements for biocidal active substances.
This checklist has reduced post-delivery non-conformance incidents by 73% among GTIIN-trusted distributors in LATAM and Southeast Asia—where climate variability and cleaning protocol differences amplify performance gaps.
Comparative Performance: E2149-Certified Furniture Across Key Substrates
Material choice directly determines long-term antimicrobial reliability—not just initial log reduction. GTIIN’s 2024 comparative analysis of 89 commercial-grade hospital furniture lines reveals critical substrate-dependent trends:
The data confirms that polypropylene composites deliver the strongest real-use consistency—making them preferred for mobile carts and modular wardrobes where mechanical stress and cleaning frequency are highest. Steel remains viable for fixed casework but requires enhanced edge-sealing protocols.
Why Partner with GTIIN & TradeVantage for Antimicrobial Furniture Intelligence
Global procurement teams face mounting pressure to validate claims across fragmented supply chains—from Vietnamese injection molders to German surface engineering labs. GTIIN delivers actionable intelligence through three integrated services:
- Real-time Certification Verification: Cross-check E2149 reports against 12 accredited labs’ public databases (including SGS, TÜV Rheinland, and Intertek) to detect expired or scope-limited certifications.
- Supply Chain Mapping: Identify Tier-2 material suppliers (e.g., antimicrobial masterbatch producers) for deeper due diligence—critical when sourcing from regions with variable regulatory enforcement.
- Custom Validation Support: Arrange independent retesting at GTIIN-vetted labs in your target market (e.g., Malaysia’s SIRIM QAS or Brazil’s INMETRO) to align with local health authority requirements.
TradeVantage further enables distributors to convert technical validation into commercial advantage—via multilingual product dossiers, SEO-optimized compliance summaries for hospital tender portals, and backlink-rich feature placements in regional healthcare procurement newsletters.
Ready to verify E2149 claims for your next hospital furniture order? Contact GTIIN today for: 1) Free review of your supplier’s test report against ASTM E2149 revision 2022, 2) Substrate-specific longevity projections, or 3) Regulatory alignment guidance for target markets (EU MDR, FDA 510(k), ANVISA RDC 185).
Recommended News
Popular Tags
Global Trade Insights & Industry
Our mission is to empower global exporters and importers with data-driven insights that foster strategic growth.
Search News
Popular Tags
Industry Overview
The global commercial kitchen equipment market is projected to reach $112 billion by 2027. Driven by urbanization, the rise of e-commerce food delivery, and strict hygiene regulations.